If you are a US HEALTHCARE PROFESSIONAL:
If you are a PATIENT or CAREGIVER:
You are viewing the PATIENTS/CAREGIVERS site.
If you are a US HEALTHCARE PROFESSIONAL:
If you are a PATIENT or CAREGIVER:
How to Administer RYALTRIS®
Administer RYALTRIS® by the intranasal (nose) route only. Do not spray RYALTRIS® into your eyes or mouth.1
Preparing the nasal spray bottle
Before you use RYALTRIS® for the first time, you will need to shake the bottle well and prime the pump.
Priming
Prime RYALTRIS® before initial use by releasing 6 sprays. When RYALTRIS® has not been used for 14 days or more, re-prime by releasing 2 sprays or until a fine mist appears.
Avoid spraying RYALTRIS® into the eyes or mouth.
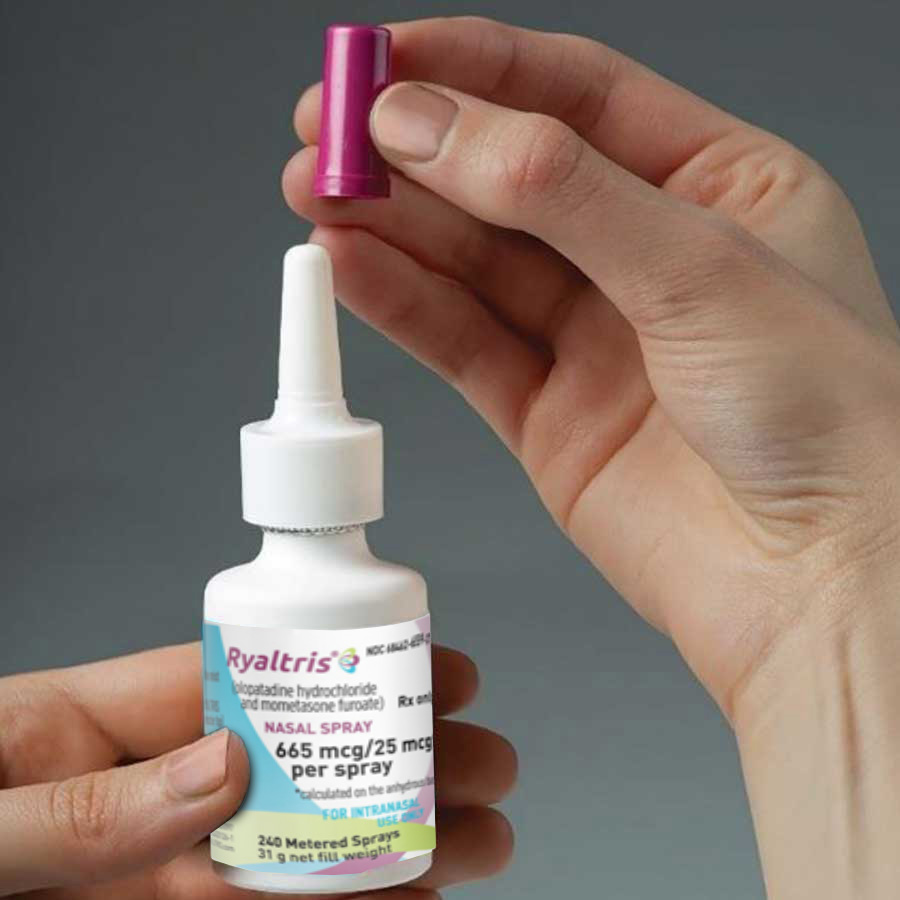
Step 1
Remove the dust cap
Remove the purple plastic dust cap from the spray pump tip of the bottle.

Step 2
Preparing the nasal spray bottle
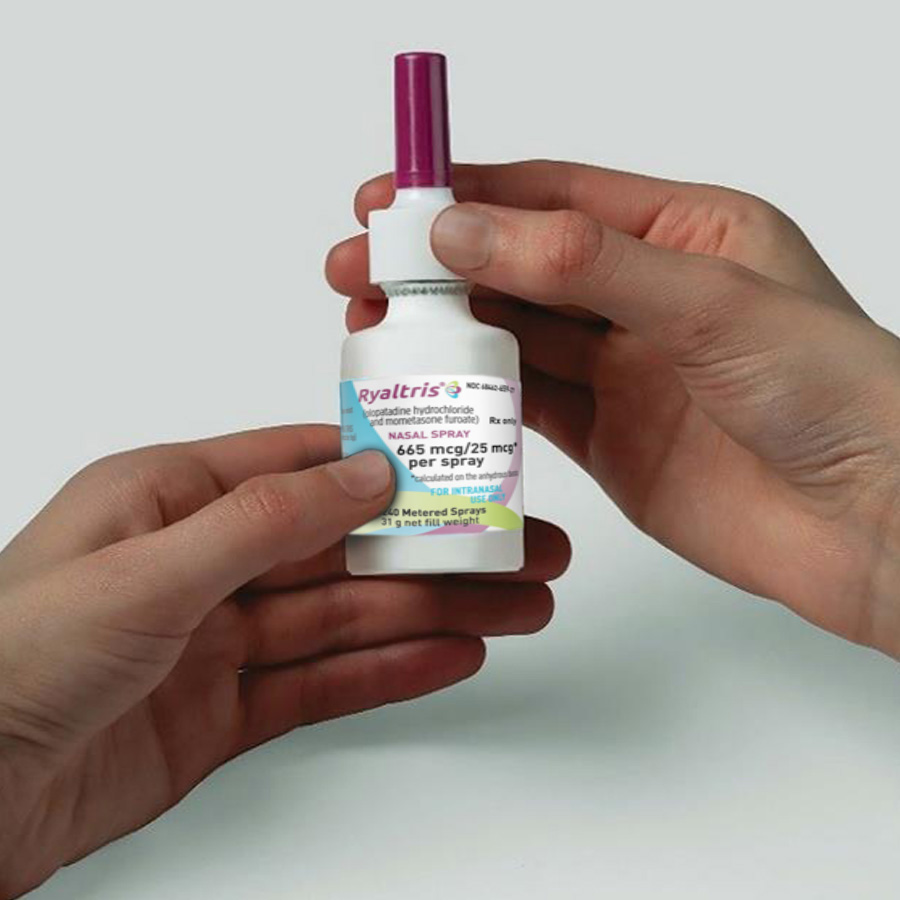
Hold the nasal spray bottle firmly and upright with your index and middle fingers on either side of the applicator (on finger rests) while supporting the grooved base of the bottle with your thumb.
Before first use, push down on the pump quickly and firmly 6 times, releasing the spray into the air, away from the eyes and face until a fine mist appears.
If you do not use RYALTRIS® for 14 or more days, you will need to shake the bottle well, and prime the pump with 2 sprays or until a fine mist appears.
RYALTRIS® is now ready for use.

Step 3
Using RYALTRIS®
Gently blow your nose to clear your nostrils.
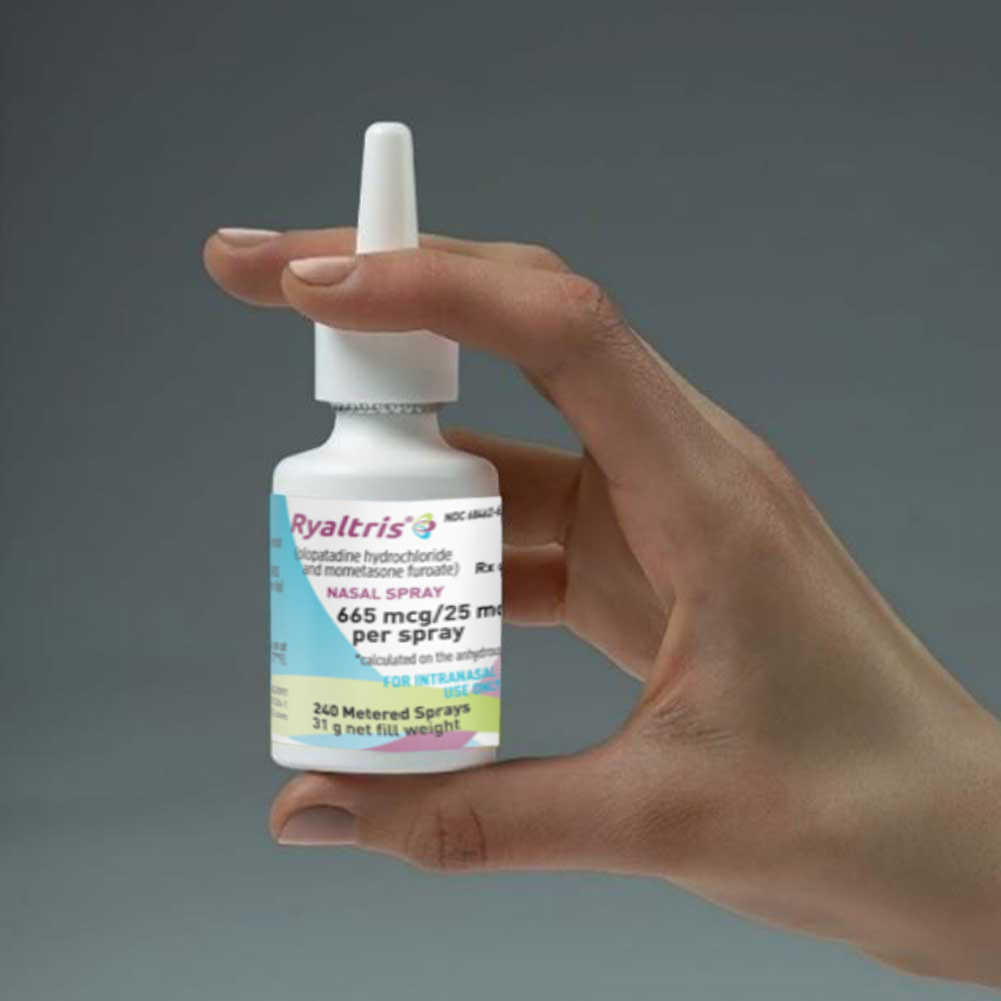
Step 4
Using the nasal spray
Shake the bottle well before each use (morning and evening).
Hold the bottle firmly with your index and middle fingers on either side of the applicator (on finger rests) while supporting the grooved base of the bottle with your thumb.

Hold 1 nostril closed with a finger. Insert the end of the spray pump tip into the other nostril, pointing it slightly toward the outside of the nose, away from the nasal septum (the wall between the 2 nostrils).

Tilt your head forward slightly. Keep the bottle upright and press down once quickly and firmly on the finger rests to activate the pump. Breathe in (inhale) gently through your nose as you spray. Then breathe out through your mouth.
Try not to get any spray in your eyes or directly on your nasal septum (the wall between the 2 nostrils).
Repeat all of step 4 and deliver a second spray in the same nostril.
Repeat all of step 4 with 2 sprays in the other nostril.
Do not blow your nose for at least 15 minutes after using RYALTRIS®, to make sure that you receive all of the medicine.
Do not tip your head back right after using to keep the medicine from going into your throat.
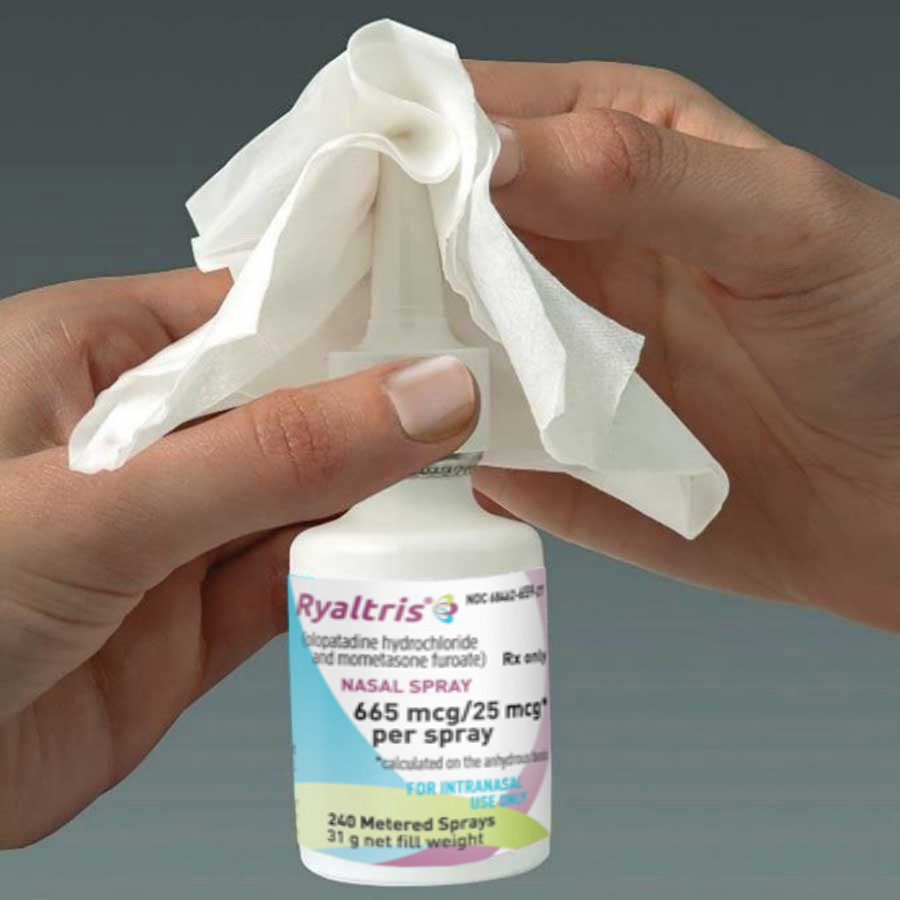
After you finish using the medicine, wipe the tip with a clean, dry tissue or cloth.
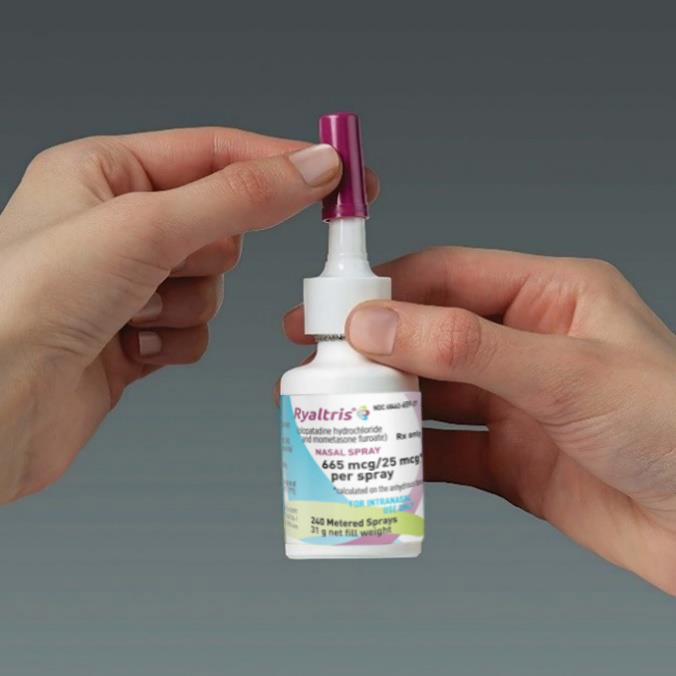
Step 5
Cleaning your bottle
Hold the spray pump unit and push the dust cap back on the spray pump tip of the bottle until you hear a click.
Each bottle of RYALTRIS® contains enough medicine for you to spray from the bottle 240 times after the first (initial) priming. You should keep track of the number of sprays used from each bottle of RYALTRIS®. Do not count any sprays used for initial priming of the bottle.

Step 6
How to clear the RYALTRIS® spray pump unit if it becomes blocked
Do not try to unblock the spray pump unit by inserting a pin or other sharp object. This will damage the spray pump unit, and you may not get the correct dose of medicine.
If the spray pump unit becomes blocked, remove it by gently pulling upward. Remove the dust cap and place only the spray pump unit in warm water to soak.
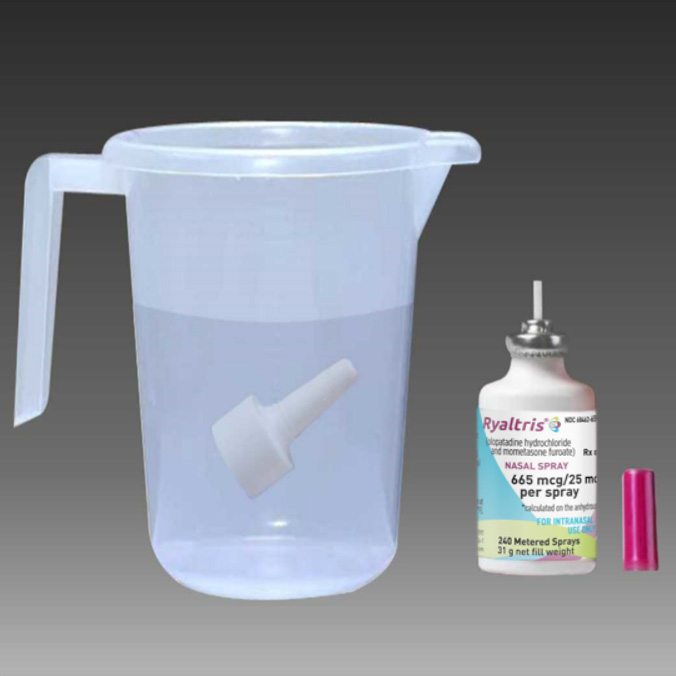
After soaking for 15 minutes, rinse the spray pump unit and dust cap with warm water, and allow them to dry completely.
Place the dust cap on the spray pump tip and put the spray pump unit back on the bottle.
After following the steps to clear your blocked spray pump see “Preparing the nasal spray bottle and priming” above and re-prime using 2 sprays. Replace the dust cap, and your RYALTRIS® is ready for use.
How should I store RYALTRIS®?1
- Store RYALTRIS® upright with the dust cap on at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not freeze or refrigerate.
- Safely throw away medicine that is out of date or no longer needed.
- Throw away your RYALTRIS® bottle after using 240 sprays after the first priming. Even though the bottle may not be completely empty, you may not get the correct dose of medicine if you continue to use it.
- Keep RYALTRIS® and all medicines out of reach of children.
General information about the safe and effective use of RYALTRIS®: Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not use RYALTRIS® for a condition for which it was not prescribed. Do not give RYALTRIS® to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about RYALTRIS® that is written for healthcare professionals.
Reference
- RYALTRIS® Nasal Spray approved prescribing information, Glenmark Specialty SA, Switzerland.

